
The effect of native yeasts on botrytis bunch rot was scored on whole Thompson Seedless berries at Washington State University in Pullman in 2015 using this disease severity rating scale. In many cases, the yeasts reduced the disease severity of a number of pathogen isolates. (Photo by Xuefei Wang)
In the future, wine grape growers may be just as interested in a particular species of native yeast as winemakers, but for vastly different reasons. Research supported by the Washington State Wine Commission shows that native yeasts of Washington vineyards have potential to help combat fungal disease of wine grapes, reduce fungicide use and ensure consistent wine quality.
Winemakers use cultured and native yeasts to turn grape juice into wine. The choice of yeast can also influence wine quality by contributing aroma, flavor and texture and inhibiting unwanted yeasts and spoilage microorganisms.
Up to this point growers haven’t put much thought into yeast populations in the vineyard, but today’s DNA sequencing technology could change that. It brings new scientific interest in native yeasts for their potential biocontrol of grape pathogens and contributions to wine quality.

Graduate student Xuefei Wang harvests Cabernet Sauvignon grape clusters in Washington’s Horse Heaven Hills in 2015 for native yeast diversity and fermentation studies. (Courtesy Patricia Okubara)
Native yeasts in Washington
Research supported by the Washington wine industry found that a number of native yeasts from Washington vineyards inhibited the growth of botrytis bunch rot, albeit under laboratory settings. The project was led by Patricia Okubara, plant geneticist for the U.S. Department of Agriculture in Pullman. Her research program studies crop plant defense responses to pathogens, disease resistance and biological control.
Okubara and graduate student Xuefei Wang collected Cabernet Sauvignon grapes from the Horse Heaven Hills and Walla Walla Valley at veraison and harvest in 2015 and 2016 and took the grapes through small-scale native fermentations, without added yeast. Through DNA sequencing, Okubara and her team identified up to 180 yeast species from both seasons.
Advances in DNA sequencing have moved this type of research from being labor-intensive, culture-based and largely manual to techniques used today that give a broad picture from hundreds of thousands of sequences, with each sequence equivalent to a single cultured yeast.
“DNA sequences are like the census,” she explained. “They’re like nametags telling us the types of yeasts present (like family members) and how many of each type are in a sample, like a head count of family members.”
Yeast is a unicellular fungus and widely distributed in nature — about 1,500 yeast species are known. They readily adapt to changes in climate, temperature and moisture. Earlier research found that some yeasts are antagonists to food and fruit pathogens.
Of the 180 species found in the two Washington vineyards, 20 were abundant, which indicated that a small number of yeasts were strongly associated with the grape berries, Okubara said. About 10 yeasts were present during the early stages of fermentation and could play a part in affecting the wine quality.
Factors that influenced yeast types found in the vineyard were location and year of sampling (conditions at the time of sampling).
“We found yeasts common in both vineyards, but also unique to each vineyard,” she said.
The yeasts did not show undesirable wine effects in the study, such as the spoilage yeast Brettanomyces. Okubara did not see much effect from sulfites on the native yeasts during fermentation, although sulfites seemed to reduce the numbers of yeast types.
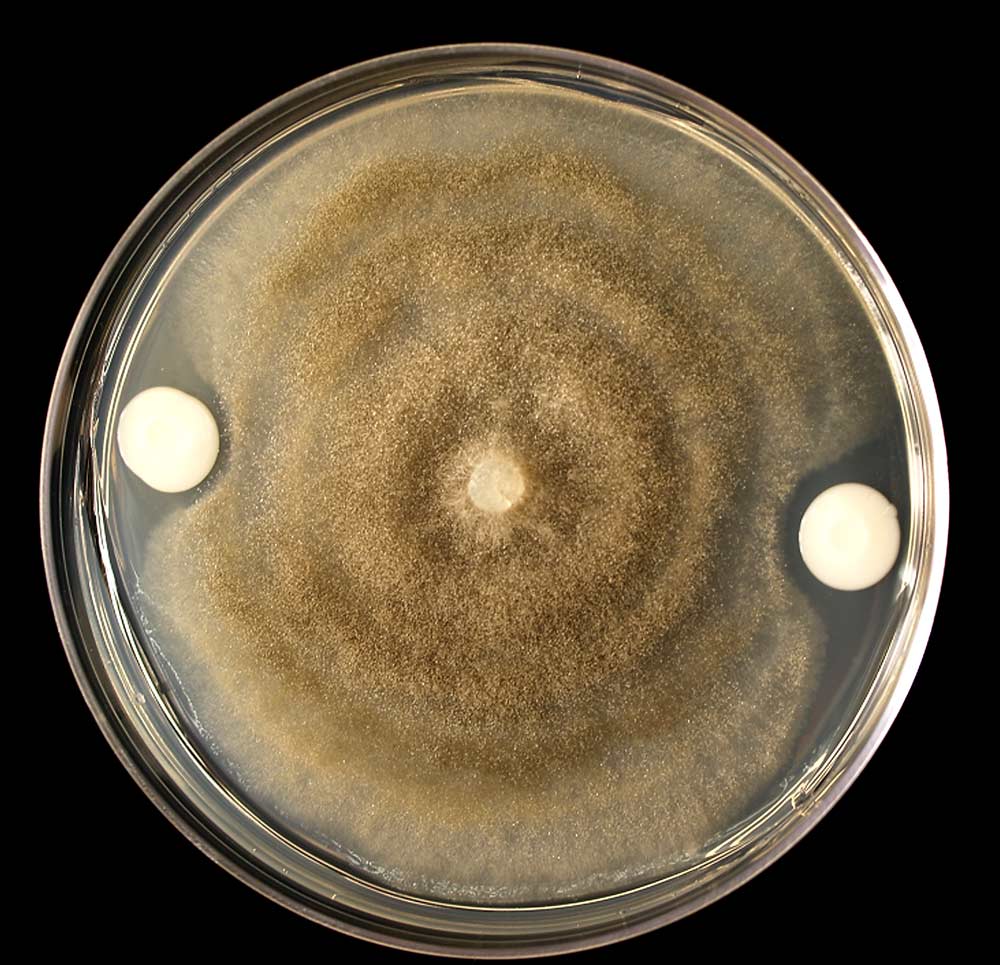
Growth inhibition of Botrytis cinerea 207b (left) in the presence of native yeast Meyerozyma guilliermondi P34D003 (right) on synthetic medium during studies at Washington State University in Pullman in 2015. The clear zone around the yeast indicates that it is producing a chemical that blocks pathogen growth. (Courtesy Xuefei Wang)
Biocontrol potential
“Native yeasts are attractive as biocontrol agents of fungal diseases because they have simple nutritional requirements, are stable under storage and rapidly colonize openings in the berry skin, flower or other parts of the vine, including wounds and early pathogen infection sites,” Okubara said. Commercial biofungicides based on yeasts already have been developed for postharvest control of Botrytis cinerea in citrus and pome fruit, she added.
In grapes, Botrytis cinerea serves as the causal agent for botrytis bunch rot, a destructive decay of grapes that reduces wine quality and impacts postharvest storage of table grapes.
Most grape cultivars are susceptible to bunch rot, also known as gray mold. Although the fungal disease is most severe in years with cool, wet growing conditions, it’s a problem worldwide for grape growers and caused millions of dollars of damage in Eastern Washington in 2010.
Management practices to control bunch rot include applying fungicides, keeping the canopy open for air movement and spray penetration, removing leaves and choosing clones with loose cluster architecture.
Okubara’s study found that six of the most abundant yeasts collected from the Washington vineyards inhibited growth of the botrytis bunch rot pathogen on grape berries in the laboratory.
And the study found another positive: Many of the yeasts were resistant to popular fungicides, making them potentially suitable for integrated pest management.
Some of the yeast strains showed partial to complete tolerance of the fungicides Pristine (pyraclostrobin, boscalid), Luna (fluopyram), Procure (triflumizole) and Vivando (metrafenone).
But just as there are diverse species of yeast, there are different isolates or strains of Botrytis. The project tested the ability of 11 native yeast species to inhibit nine different Botrytis pathogens; each pathogen responded differently to each yeast.
“No single yeast was effective against all Botrytis pathogens,” Okubara said. “This indicated that the biocontrol response depends both on the particular yeast and particular pathogen.”
Future
Okubara believes that native yeasts could out-compete the Botrytis pathogen if they could colonize the berry earlier. Niche competition is a common mechanism of biocontrol.
Her study results suggest the possibility of combining yeasts for use in the vineyard, but due to the complexities of the yeast-pathogen interactions, further research is required before native biocontrol yeasts can be tested for use in the vineyard.
She envisions a day when certain yeasts could serve as indicators of vineyard health. A quick lab test could analyze grower samples and identify the types of yeasts populating the vineyard.
Research is underway to identify connections between yeasts and environment or management practices, but much more research is needed to expand the study of yeast species in Washington vineyards, identify those that provide biocontrol of bunch rot and better understand what growers can do to enhance beneficial yeasts in the vineyard. Okubara’s research is a first step toward ultimate commercialization of yeasts as biocontrol agents for bunch rot. •
Melissa Hansen is the research program manager for the Washington State Wine Commission. A detailed report of Okubara’s research is available on the research page of the commission’s website at washingtonwine.org.






Leave A Comment